New publication examines the role of organic acids in controlling siderite oxidation
Low molecular weight organic acids stabilise siderite against oxidation and influence the composition of iron (oxyhydr)oxide oxidation products: This is the title and major finding of the new article has rescently been published in the journal Environmental Science Processes and Impacts.
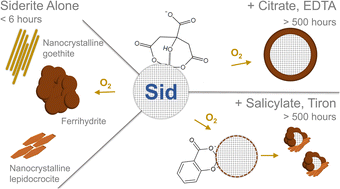
Low molecular weight organic acids stabilise siderite against oxidation and influence the composition of iron (oxyhydr)oxide oxidation products
Siderite (FeCO3) is an important reservoir of mineral-bound ferrous iron in non-sulfidic, reducing soils and sediments. It is redox sensitive, and its oxidation may facilitate the reduction of a range of pollutants, produce reactive oxygen species, or induce the formation of oxidation products with large surface areas for contaminant sorption. However, there is currently a limited understanding of the stability of siderite in complex environments such as soils and sediments. Here, we use a series of batch experiments complemented with thorough characterisation of mineral oxidation products to investigate the oxidation of siderite in the presence and absence of the low molecular weight organic acids (LMWOAs) citrate, tiron, salicylate, and EDTA as analogues for naturally occurring compounds or functional groups of natural organic matter that ubiquitously coexist with siderite. Our results show that siderite alone at pH 7.5 was completely oxidised to form ferrihydrite, nanocrystalline lepidocrocite, and nanocrystalline goethite in less than 6 hours. However, in the presence of LMWOAs, up to 48% of the siderite was preserved for more than 500 hours and the formation of goethite was inhibited in favour of ferrihydrite and lepidocrocite. Using experimental data from electron microscopy and chemical speciation modelling, we hypothesise that the siderite may be preserved through the formation of an Fe(III)-passivation layer at the siderite surface.